Does ChemoCentryx Have a Platform Problem?
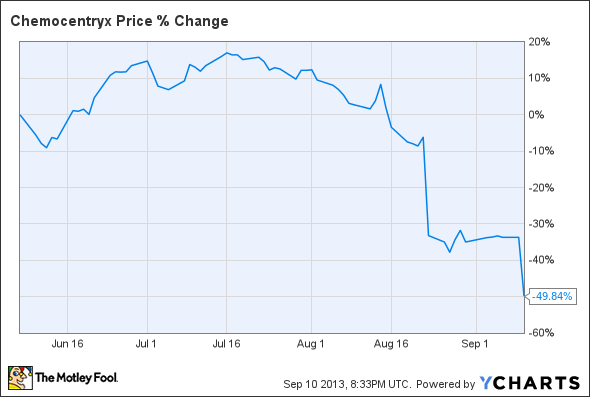
It hasn't been a fun month to be an investor in clinical-stage biotech ChemoCentryx . After announcing at the end of August that its lead drug candidate vercirnon failed the first of four late-stage trials targeting Crohn's disease, the company dropped a bomb Tuesday with less-than-riveting top-line interim results for its second most advanced drug candidate. CCX140 is being evaluated in a phase 2 trial in patients with diabetic nephropathy, or diabetic kidney disease. Results failed to impress some analysts and shares finished the day down more than 24%. They have been sliced in half in the last three months alone.
Investors could look at recent developments as a short-term hiccup in the road for a developmental company, but I may be a little more cautious about investing in ChemoCentryx. Why? The company has positioned its entire pipeline to take advantage of a specific biological pathway shown to be associated with inflammation. Should two early misses with its most advanced drugs push investors toward the exits?
#platformproblems
The company's platform is centered on inhibiting the chemokine system, which is a complex communication system between cell receptors and the immune system. When certain signal-inducing cells aggregate at a source of inflammation a specific immune response is triggered. Inhibiting receptors has been shown to reduce inflammation, but will it do so with significance in a clinical setting? That is the question investors need to begin asking themselves.
In case you have been asleep in the past, well, decade or so, anti-inflammatory drugs are hot commodities in biotech. Companies have had the most success inhibiting the TNF-alpha pathway, while JAK inhibitors are quickly shifting from potential to performance. Consider how AbbVie and Johnson & Johnson have fared with their first-generation immunology drugs:
Drug or Sector | 1H13 Sales | Year-Over-Year Growth |
|---|---|---|
Remicade | $3.27 billion | 7.5% |
Immunology, JNJ | $4.45 billion | 16.5% |
Humira | $4.85 billion | 13.9% |
Source: SEC filings.
Humira brings in more than half of AbbVie's total revenue and will become just the second drug ever to record $10 billion in a single year. That poses potential problems down the road, but investors sure aren't complaining about owning a piece of the most successful drug in the world. All companies want to lead their markets and AbbVie has done exactly that. And while Johnson & Johnson relies heavily on Remicade, it has already begun transitioning its anti-inflammatory portfolio to expanded treatment options for Simponi and its next-generation blockbuster Stelara. In fact, Stelara has enjoyed one of the most successful launches in the history of the industry on the heels of incredible convenience for patients: Far fewer injections are given in the same time period when compared to previous generation blockbusters.
What's the point? When companies develop novel anti-inflammatory drugs, they look up and see Humira and Remicade. It's a great commercial path to pursue. The idea of getting in on the ground level of the next big inflammation treatment pushed GlaxoSmithKline to partner with ChemoCentryx in 2006. The two are developing drugs against four targets, including the receptor CCR9 with vercirnon. Although the lead drug failed its first late-stage trial, the pair has had success elsewhere. Glaxo is now financially responsible for developing CCX354 for rheumatoid arthritis after positive phase 2 data. Additionally, the company is expected to make a decision on its option for CCX168 for treating ANCA vasculitis by the end of this year. Will recent developments affect that decision?
A silver lining?
There are a total of 10 drug programs ranging from preclinical to late-stage development at ChemoCentryx. All target a unique chemokine receptor. I don't know how differently each works, but if I may leverage my understanding of other anti-inflammatory drugs targeting various interleukin, JAK, or TNF-alpha receptors, then it seems entirely plausible that inhibiting some receptors will prove more clinically beneficial than other seemingly similar targets. Additionally, the failed phase 3 trial with GlaxoSmithKline was just one of four for vercirnon and the CCX140 trial is still ongoing and has yet to officially be labeled as a failure.
Foolish bottom line
Whatever stance you take with recent developments at ChemoCentryx, I would certainly begin to play devil's advocate at an increasing rate. If there is any chance that targeting the chemokine system as an effective means of managing inflammation doesn't work, then the risk-to-reward balance certainly wouldn't support an investment. There is still potential for any one of its clinical or preclinical programs to work, I suppose, but I believe there are better investments out there.
Perhaps ChemoCentryx snags a winning anti-inflammatory pathway, but its platform focus is beginning to worry me given the latest results. Luckily, the biotech industry has been soaring recently. Even then, the best investment strategy is to pick great companies and stick with them for the long term. The Motley Fool's free report "3 Stocks That Will Help You Retire Rich" not only shares stocks that could help you build long-term wealth but also winning strategies that every investor should know. Click here to grab your free copy today.
The article Does ChemoCentryx Have a Platform Problem? originally appeared on Fool.com.
Fool contributor Maxx Chatsko has no position in any stocks mentioned. Check out his personal portfolio, his CAPS page, or follow him on Twitter @BlacknGoldFool to keep up with his writing on energy, bioprocessing, and biotechnology.The Motley Fool recommends and owns shares of Johnson & Johnson. Try any of our Foolish newsletter services free for 30 days. We Fools may not all hold the same opinions, but we all believe that considering a diverse range of insights makes us better investors. The Motley Fool has a disclosure policy.
Copyright © 1995 - 2013 The Motley Fool, LLC. All rights reserved. The Motley Fool has a disclosure policy.