How High Can Ariad Fly?
Shares of Ariad Pharmaceuticals (NAS: ARIA) hit a 52-week high today. Let's look at how the company got there to find out whether clear skies remain on the horizon.
How it got here
A drug approval wasn't in the cards for Ariad and partner Merck (NYS: MRK) earlier this month, but that hasn't mattered to the market. Investors remain more interested in ponatinib, Ariad's as-yet-unapproved leukemia treatment, than they were in the rejected sarcoma treatment ridaforolimus. Since Ariad is the sole owner of ponatinib, that drug's potential presents a much more appealing long-term revenue stream than one otherwise split with Merck or another partner.
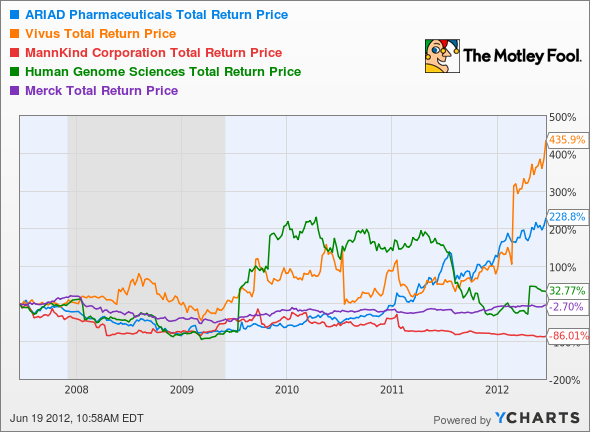
Investors, including legendary currency trader George Soros, are willing to wait for a blockbuster. Soros recently added both Ariad and Vivus (NAS: VVUS) , another development-stage biotech, to his holdings. Analysts from Summer Street Research also presented a bull case for Ariad earlier this year. In the wild world of development-stage biotech, that sense of optimism can be more than enough to generate steady growth, as opposed to Vivus' big bounce earlier this year, or Human Genome Sciences' (NAS: HGSI) roller-coaster ride:
ARIA Total Return Price data by YCharts
Ariad's big bounce may be yet to come, but that's still just over the horizon. Let's look at what's happening right now with Ariad and its peers, to see whether its price is justified.
What you need to know
There's not much in the way of revenue or profit to speak of, so let's focus on what we know about these emerging biotechs, particularly their product pipelines and the financial commitment they've made to see the drugs in those pipelines through.
Company | Drugs in Development Pipeline | Average Analyst Share-Price Target | TTM R&D Spending |
|---|---|---|---|
Ariad | 4 | $19.13 | $89 million |
Vivus | 1 | $30.18 | $26 million |
MannKind (NAS: MNKD) | 4 | $5.20 | $93 million |
Human Genome Sciences (NAS: HGSI) | 7 | $13.64 | $151 million |
Sources: Yahoo! Finance, Morningstar, and S&P Capital IQ.
Each company has its blockbuster in waiting. Ariad has ponatinib; Vivus has its anti-obesity treatment Qnexa, which already received a glowing 20-2 recommendation for approval from an FDA advisory committee; MannKind is working on inhaled-insulin treatment Afrezza; and Human Genome Sciences has lupus treatment Benlysta, which seems to be so appealing that it's attracted a buyout offer from partner GlaxoSmithKline. Each of these drugs should reach approval (or rejection) stage within a year at the most, so it may not take too long for investors to find out just how valuable they really are.
With big-time investors on its side, Ariad has the momentum today to keep growing, but there's always a risk of rejection for ponatinib waiting to send the stock tumbling. Early results look quite promising, so it's worth keeping an eye on this stock (and the company behind it, of course) to make sure that promise can be met.
What's next?
Where does Ariad go from here? You'll have to ask the FDA. Until ponatinib makes its way through the approvals process, there's not much else investors can do. The Motley Fool's CAPS community has given Ariad a lowly two-star rating, but many CAPS players see Ariad as a short-term play and are afraid to make a call for the long haul.
Interested in tracking this stock as it continues on its path? Add Ariad to your Watchlist now for all the news we Fools can find, delivered to your inbox as it happens. If you're looking for another medical stock with massive potential, you should be very interested in Motley Fool founder David Gardner's pick for "The Next Rule-Breaking Multibagger." Find out everything you need to know in our free report -- get your copy while it lasts.
At the time thisarticle was published Fool contributorAlex Planesholds no financial position in any company mentioned here. Add him onGoogle+or follow him on Twitter,@TMFBiggles, for more news and insights. The Motley Fool has adisclosure policy. We Fools don't all hold the same opinions, but we all believe thatconsidering a diverse range of insightsmakes us better investors. Try any of our Foolish newsletter servicesfree for 30 days.
Copyright © 1995 - 2012 The Motley Fool, LLC. All rights reserved. The Motley Fool has a disclosure policy.