How High Can Medivation Fly?
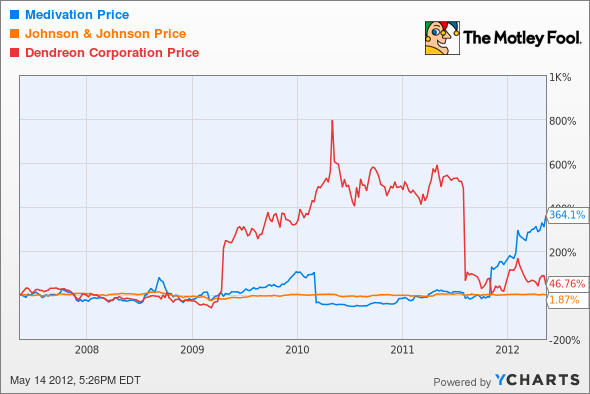
Shares of Medivation (NAS: MDVN) hit a 52-week high on Monday. Let's take a look at how the company got there and whether clear skies are still in the forecast.
How it got here
Medivation shareholders have bet the boat on enzalutamide (formerly MDV3100), the company's late-stage treatment for prostate cancer for those who have previously been treated with chemotherapy, which it co-developed with Astellas Pharmaceuticals (OTC: ALPMY).
Thus far, the data looks promising. Enzalutamide has shown statistically significant results versus a placebo test in extended survival rates among patients with very limited side effects (18.4 months versus 13.6 months for the placebo).
Johnson & Johnson-owned (NYS: JNJ) subsidiary Jaansen Pharmaceuticals reported in March that its competing drug, Zytiga, demonstrated statistical benefits in its latest study, showing a survival rate of just 4.8 months versus the placebo's 3.9 months. While the trial was unblinded because of Zytiga's effectiveness, this difference could be due to something as simple as population makeup.
Enzalutamide also has created a push-pull effect on Dendreon's (NYS: DNDN) late-stage treatment, Provenge. Because of Zytiga's results, which revealed it was effective in other stages of the disease, it now appears Dendreon could be facing possible competition from Zytiga. But let's also not forget that enzalutamide's effectiveness may be strengthened if paired with Dendreon's Provenge in the future, providing a big boost to both companies' future sales.
How it stacks up
Let's see how Medivation stacks up next to its peers.
Aside from the stable Johnson & Johnson, you can see just how hit-or-miss these biotechs can be when their pipeline consists of just a few select therapies.
Company | Price/Book | Price/Cash Flow | Forward P/E | Cash/Debt |
|---|---|---|---|---|
Medivation | 38.9 | N/M | N/M | $380 million / $187 million |
Johnson & Johnson | 2.9 | 12 | 11.7 | $33.9 billion / $19.5 billion |
Dendreon | 5 | N/M | N/M | $498 million / $574 million |
Sources: Morningstar, Yahoo! Finance. N/M = not meaningful.
Yes, I'm aware how fruitless it is to try to compare Johnson & Johnson -- a company with a huge pipeline and $14.4 billion in net cash -- to two other significantly smaller companies of which only one has a treatment currently approved by the FDA. But this exercise also helps undercover two key aspects about these prostate-cancer-fighting stocks.
First, it demonstrates that all three are very well capitalized. Clearly, Johnson & Johnson is, but Medivation is in exceptionally good shape with $193 million in net cash heading into a key year in which it will submit a new drug application to the FDA. Even Dendreon, which has struggled miserably in launching Provenge, has ample cash to ramp up its marketing campaign and weather losses as the treatment gains acceptance with physicians.
Secondly, it reinforces the fact that there are plenty of obscure drug producers waiting in the wings that aren't publicly traded, so our guard as investors must always be up. In addition, these companies must always remain innovative. According to a recent preclinical study whose findings were made public in a Cancer Research report, enzalutamide's developers created several other androgen receptor compounds, including ARN-509, owned by private-held Aragon, which may prove more effective than enzalutamide.
What's next
Now for the real question: What's next for Medivation? The answer to that loaded question really depends on whether enzalutamide is approved by the FDA, whether it can successfully launch the drug if approved, and whether it can stand up to the fierce competition among its peers -- both public and private.
Our very own CAPS community gives the company a two-star rating (out of five), with 71.1% of members expecting it to outperform. I have made a CAPScall of underperform on Medivation and thus far it remains one of my biggest goofs to date, currently down a shameful 103 points. However, I figure I've come this far, so why stop now?
Over the past year, we've seen a mixture of failures and successes on drug launches. Dendreon failed to take into account the fact that Provenge's price ($93,000) would cause physicians to shy away from prescribing the treatment. On the flip side, Regeneron Pharmaceuticals (NAS: REGN) has educated the sector on how to properly launch a drug by undercutting the price of its closest competitor and upping the sales guidance of its wet macular degeneration treatment twice in the past few months. Somehow, I'm not convinced the price of enzalutamide will be anywhere near "reasonable" territory, and I'm concerned it may not be profitable for quite some time. Drug approval aside, Medivation has a lot to prove to investors.
Medivation clearly has a pipeline that's looking to change lives. If you'd like the inside scoop on a stock our Motley Fool Rule Breakers team feels could offer the next revolutionary product, then click here for your free access to your latest report.
Craving more input on Medivation? Start by adding it to your free and personalized watchlist. It's a free service from The Motley Fool to keep you up to date on the stocks you care about most.
At the time thisarticle was published Fool contributor Sean Williams has no material interest in any companies mentioned in this article. You can follow him on CAPS under the screen name TMFUltraLong, track every pick he makes under the screen name TrackUltraLong, and check him out on Twitter, where he goes by the handle @TMFUltraLong.The Motley Fool owns shares of Johnson & Johnson and Dendreon. Motley Fool newsletter services have recommended buying shares of and creating a diagonal call position in Johnson & Johnson. Try any of our Foolish newsletter services free for 30 days. We Fools don't all hold the same opinions, but we all believe that considering a diverse range of insights makes us better investors. The Motley Fool has a disclosure policy.
Copyright © 1995 - 2012 The Motley Fool, LLC. All rights reserved. The Motley Fool has a disclosure policy.